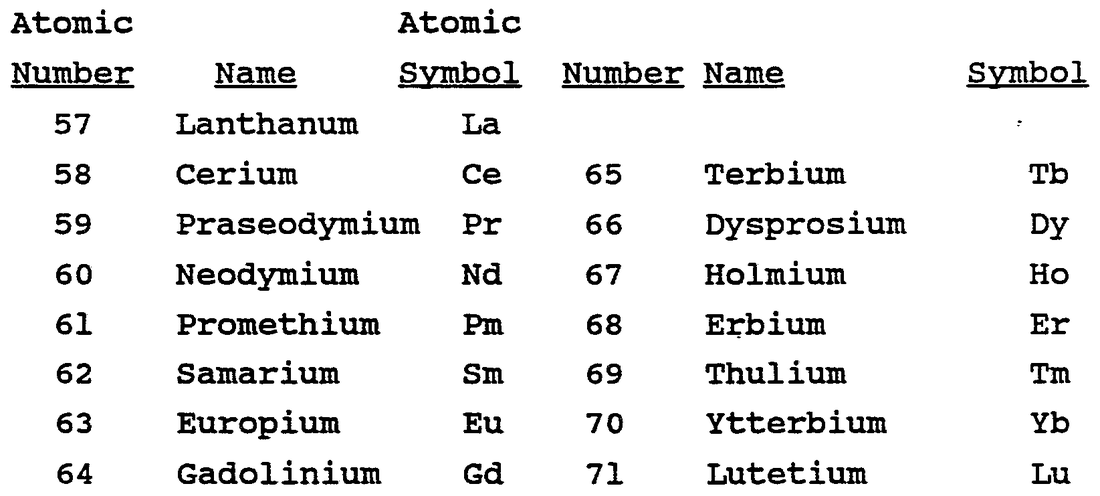
The Lanthanide Series are 15 rare-earth metals because they were believed to be rare when first discovered. They are not rare. Only one is, Promethium. It is actually radioactive. It used to be on Earth but has decayed into other elements now. The atomic numbers of Lanthanides are from 57 to 71.

Properties of these Elements: (picture of Lanthanide oxides)
- They exhibit chemical properties similar to Lanthanum.
- They are bright and silvery in appearance
- They tarnish when exposed to air.
- They are hard to differentiate from each other.
- They are also hard to separate from each other.
- They are relatively soft metals. (Some can just be cut with a knife)
- They are really reactive, reacting well with water, and dissolving in acid.
- They also have high electrical conductivity.
Uses of the Lanthanides
An early use for a Lanthanide was a flint type lighter in gas and cigarette lighters. The Lanthanides have many uses but are expensive and hard to separate from their ores and companies try to use less expensive alternatives. However, Lanthanides are great catalysts so the petroleum industry uses them to help convert crude to gasoline. They are used in street lights, search lights and in the big high intensity lights at sports stadiums. We also use them in laptops, cellphones, plasmas screen televisions, solar panels, and pollution controls, but they also create toxic waste. In 2010, China was responsible for 95% of the world's production using Lanthanides.
An early use for a Lanthanide was a flint type lighter in gas and cigarette lighters. The Lanthanides have many uses but are expensive and hard to separate from their ores and companies try to use less expensive alternatives. However, Lanthanides are great catalysts so the petroleum industry uses them to help convert crude to gasoline. They are used in street lights, search lights and in the big high intensity lights at sports stadiums. We also use them in laptops, cellphones, plasmas screen televisions, solar panels, and pollution controls, but they also create toxic waste. In 2010, China was responsible for 95% of the world's production using Lanthanides.
Some Other Cool Information About the Lanthanides!!
This video from the Discovery Channel explains the Lanthanides pretty well.
This video shows you REALLY cool reactions of Lanthanides. (I needed a little of it explained to me though.)
This Video IS AWESOME. It has so much information but BEWARE he uses the words a$$ twice, bleeps out something else and says frickin' too. (My mom made me put in this disclaimer)